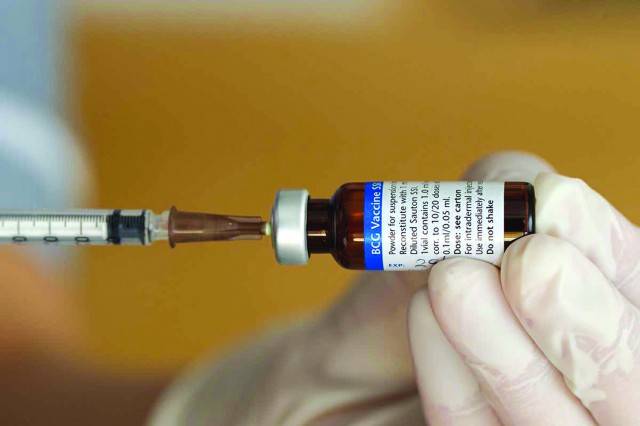
The promise of newly developed vaccines against COVID-19 has provided tremendous hope for the developed world, especially when it comes to protecting those at risk due to their advanced age. At the same time, there is growing concern for the elderly in developing countries, the vast majority of whom are not expected to get the new vaccines until at least 2022.
However, recent developments suggest that Bacille Calmette-Guérin (BCG) - an affordable, safe, and easy to transport 100-year-old vaccine used against Tuberculosis - may become a stopgap measure against COVID-19. The results of an ongoing clinical trial in the Netherlands, BCG PRIME, in which the medicine is being tested against COVID-19 on nearly 5,000 elderly people with pre-existing medical conditions, are expected by the end of January.
Initial epidemiological data was released in March 2020 and it showed a significant correlation between a nation’s universal BCG vaccination policy and low mortality per capita due to COVID-19. In April, the World Health Organization (WHO) warned against using BCG for COVID-19 before clinical trials confirmed its effectiveness. The WHO was concerned about supply shortages that would affect roughly 125 million children around the world who are inoculated with BCG at birth every year, mostly in developing countries with high incidence of Tuberculosis.
If the BCG PRIME trial shows significant reduction in infection and mortality rates amongst this most vulnerable population, it will prove this correlation: that BCG induces a protective immune response against COVID-19.
If the results of the BCG PRIME trial demonstrate a high degree of effectiveness in combatting COVID-19, scaling BCG production should not be too challenging, given that more than 500 million doses of BCG are already produced and sold at an average cost of less than $1 per dose by over 20 manufacturers around the world, including the Serum Institute of India, the world’s largest manufacturer of vaccines.
This optimism about BCG stems from the recently published results of a retrospective study done with healthcare workers in Los Angeles as well as a clinical trial in Greece, which looked at the impact of BCG on elderly patients’ post-surgery. These results build on the body of evidence collected over several decades about the unanticipated immune-boosting benefits of BCG, as evidenced by its approval by the US Food and Drug Administration (FDA) in 1990 as the first immunotherapy approved for treatment of bladder cancer.
On November 19, 2020, the Cedars-Sinai Medical Center in Los Angeles announced that previously BCG-vaccinated individuals, mostly immigrants to the US, had roughly 30 percent lower COVID-19 infection rates when compared to the cohort with no prior BCG vaccination, mostly local-born Americans, despite having a 34 percent higher rate of co-morbidities.
This study, led by Dr. Moshe Arditi, sheds light on the biggest debate in medical circles around BCG’s impact on COVID-19: whether it is durable enough to protect people for several decades and possibly even a century. Observing 30 percent lower infection rates among the group of BCG vaccinated healthcare workers, with the average age of 43, indicates the possibility of long-term protection against COVID-19. It should be noted that there were no deaths, so the possible impact of BCG against mortality due to COVID-19 could not be proven in this retrospective study of mostly young and healthy medical staff.
The benefits of BCG on the elderly were confirmed in October when results of the three-year BCG ACTIVATE clinical trial in Greece were published in Cell. When given to elderly patients upon discharge from hospital post-surgery, BCG reduced respiratory infections by 79 percent compared to the cohort that received a placebo. Regardless of how effective BCG is proven to be against COVID-19 through the BCG PRIME trial, the drug would appear to complement any COVID-19 vaccine for all the elderly in the world.
The BCG PRIME trial was launched in early September 2020 by Dr Mihai Netea, who also led the BCG ACTIVATE trial. Dr Netea coined the term “trained immunity” with his colleagues back in 2011 to explain how BCG helps build stronger immunity against many more pathogens than Tuberculosis. Dr Netea also recently co-authored an article in The New England Journal of Medicine, “Trained Innate Immunity, Epigenetics, and COVID-19,” where he laid out the theory behind how BCG may be providing protection against COVID-19.
On December 11, the BCG PRIME team announced at the International Symposium for Innate Immunity and COVID-19 that they expected results by the end of January.
If BCG is proven to be effective against COVID-19, it is imperative to address the notion that BCG is what is protecting the elderly in countries with high rates of inoculation.
What really matters in the case of COVID-19 is whether the elderly in a country, i.e. those over 70 years of age, are protected or not. The countries where we found strong evidence that a majority or supermajority of the over-70 population have received BCG include Greece, Norway, Finland, Denmark, Germany, New Zealand, Australia, Japan, Vietnam and Uruguay. These countries have very low death rates per capita due to COVID-19, far lower than those observed in the US, Italy, Belgium, and the Netherlands, the only four nations in the world to have never adopted BCG.
In fact, Belgium has the highest COVID-19 death rate per capita of any country in the world, though it ranks 17th among all nations on the United Nation’s Human Development Index (HDI). Even more remarkable is that Greece, with a similar sized population to Belgium, has a death rate per capita that is one quarter that of Belgium, despite being ranked 32nd on the HDI, and with a far weaker healthcare system. Greece introduced BCG in 1925 and by the early 1950s had vaccinated a vast majority of the children under the age of 18, meaning a supermajority of the current 70+ population born in Greece is BCG vaccinated compared to almost no local born person over age 70 vaccinated in Belgium.
Japan provides another useful example of a country where the correlation appears to indicate that there is likely causality. Having initially introduced BCG in 1924, Japan decided in 1949 to vaccinate all citizens under 30. Thus, we expect that almost everyone between 70-101 years of age in Japan has been vaccinated with BCG. This may explain why Japan, a country of 126 million people, with 27 million over the age of 70, has lost only 3,300 of its citizens due to COVID-19 in 2020, despite Japan’s initial “half-hearted” response to the pandemic.
All of the other countries that have high death rates per capita (UK, France, Spain, Brazil, Mexico, Chile) are those where we did not find any evidence of a majority of the population over 70 having BCG protection.
If the BCG PRIME trial shows proof of immunity against COVID-19, then critical decisions will need to be made to determine how to prioritize the new supply of BCG which we expect to be produced by the 20+ existing global manufacturers at a rapid pace.
When analyzing the effectiveness of BCG over a lifetime, it was observed in a clinical trial conducted between 1935 and 1998 among almost 3,000 indigenous American and Alaskan peoples that those who received BCG between 1935 and 1938 had a 60 percent lower chance of developing lung cancer 60 years later as adults, compared to those in the control placebo group, highlighting the possible long-term benefits of BCG.
While our research did not find evidence of high BCG coverage for the over-70 population in any Latin American country other than Uruguay, there are a large number of countries in Asia and Africa that have fared well during the COVID-19 crisis, even without heavy BCG coverage in the over 70 population, such as Pakistan, Malaysia, Nigeria, and Egypt.
While more research and trials need to be conducted to better understand why so many of these countries where the elderly are not covered with BCG tend to have very low mortality rates, our analysis leads us to attribute the positive outcomes in these countries to a high level of Latent Tuberculosis among the elderly.
Several recent studies, including an instrumental variable method analysis from Japan, highlight a strong correlation between low death rates from COVID-19 and high levels of Latent Tuberculosis in most Asian and African countries. According to these studies, Latin America has a much lower incidence of Latent Tuberculosis than Asia and Africa, closer to that of North America and Western Europe.

Our research also brought up an astonishing statistical disparity within the United States, where 38 percent of the roughly 378,000 deaths have taken place in nursing homes despite reflecting only five percent of cases. Homeless shelters, on the other hand, have been spared despite high levels of infections. In fact, in eight homeless shelters across the country with 2,665 residents, 778 people (29 percent) tested positive for COVID-19, yet not a single person died. In three of those homeless shelters, all 214 infected patients were asymptomatic, a near statistical impossibility given that an estimated 45 percent of COVID-19 patients are asymptomatic.
The US Center for Disease Controls (CDC) was reported to be actively looking into the “stunning” results from the Pine Street Inn Homeless shelter in Boston back in mid-April. While Latent Tuberculosis is quite rare in the US, prevalence of Latent TB in the homeless population in the United States has been reported to be as high as 67 percent, an incidence rate 20 times higher than the general population.
In his book, Discovering Tuberculosis, Christian McMillen writes that “the mass vaccination with BCG, is without doubt, the most far reaching and longest-lasting prophylactic medical campaign in world history.” According to McMillen, BCG, which was developed by Albert Calmette and Camille Guérin in 1921 at the Pasteur Institute in France, has been given to an estimated 4.5 billion people around the world, and another 1.8 billion carry Latent Tuberculosis, roughly 20x more people than those living with Active Tuberculosis disease. That means up to 6.3 billion people in the world could have the Tuberculosis germ either through the live attenuated Tuberculosis in the BCG vaccine or by virtue of infection with Latent Tuberculosis.
Our research and analysis suggest that countries with high levels of BCG vaccination and/or high levels of Latent Tuberculosis among the elderly have significantly lower mortality rates than those without. Many developed nations already have the new vaccines against COVID-19, some with contracts ample to vaccinate their populations several times over. If the results of the BCG PRIME trial are positive, the elderly in many developing countries, especially those in Latin America, who remain most at risk of COVID-19 fatality, should be prioritized in order to have a more equitable BCG rollout than has been the distribution of newly developed COVID-19 vaccines.
Kashif Zafar is a financial services executive based in New York and serves on the boards of the American Pakistan Foundation and Middlebury College.
Adil Alvi is a first year student at Middlebury College having graduated from The Lyceum in Karachi, Pakistan
However, recent developments suggest that Bacille Calmette-Guérin (BCG) - an affordable, safe, and easy to transport 100-year-old vaccine used against Tuberculosis - may become a stopgap measure against COVID-19. The results of an ongoing clinical trial in the Netherlands, BCG PRIME, in which the medicine is being tested against COVID-19 on nearly 5,000 elderly people with pre-existing medical conditions, are expected by the end of January.
Initial epidemiological data was released in March 2020 and it showed a significant correlation between a nation’s universal BCG vaccination policy and low mortality per capita due to COVID-19. In April, the World Health Organization (WHO) warned against using BCG for COVID-19 before clinical trials confirmed its effectiveness. The WHO was concerned about supply shortages that would affect roughly 125 million children around the world who are inoculated with BCG at birth every year, mostly in developing countries with high incidence of Tuberculosis.
If the BCG PRIME trial shows significant reduction in infection and mortality rates amongst this most vulnerable population, it will prove this correlation: that BCG induces a protective immune response against COVID-19.
If the results of the BCG PRIME trial demonstrate a high degree of effectiveness in combatting COVID-19, scaling BCG production should not be too challenging, given that more than 500 million doses of BCG are already produced and sold at an average cost of less than $1 per dose by over 20 manufacturers around the world, including the Serum Institute of India, the world’s largest manufacturer of vaccines.

This optimism about BCG stems from the recently published results of a retrospective study done with healthcare workers in Los Angeles as well as a clinical trial in Greece, which looked at the impact of BCG on elderly patients’ post-surgery. These results build on the body of evidence collected over several decades about the unanticipated immune-boosting benefits of BCG, as evidenced by its approval by the US Food and Drug Administration (FDA) in 1990 as the first immunotherapy approved for treatment of bladder cancer.
On November 19, 2020, the Cedars-Sinai Medical Center in Los Angeles announced that previously BCG-vaccinated individuals, mostly immigrants to the US, had roughly 30 percent lower COVID-19 infection rates when compared to the cohort with no prior BCG vaccination, mostly local-born Americans, despite having a 34 percent higher rate of co-morbidities.
Optimism about BCG stems from the recently published results of a retrospective study done with healthcare workers in Los Angeles as well as
a clinical trial in Greece
This study, led by Dr. Moshe Arditi, sheds light on the biggest debate in medical circles around BCG’s impact on COVID-19: whether it is durable enough to protect people for several decades and possibly even a century. Observing 30 percent lower infection rates among the group of BCG vaccinated healthcare workers, with the average age of 43, indicates the possibility of long-term protection against COVID-19. It should be noted that there were no deaths, so the possible impact of BCG against mortality due to COVID-19 could not be proven in this retrospective study of mostly young and healthy medical staff.
The benefits of BCG on the elderly were confirmed in October when results of the three-year BCG ACTIVATE clinical trial in Greece were published in Cell. When given to elderly patients upon discharge from hospital post-surgery, BCG reduced respiratory infections by 79 percent compared to the cohort that received a placebo. Regardless of how effective BCG is proven to be against COVID-19 through the BCG PRIME trial, the drug would appear to complement any COVID-19 vaccine for all the elderly in the world.
The BCG PRIME trial was launched in early September 2020 by Dr Mihai Netea, who also led the BCG ACTIVATE trial. Dr Netea coined the term “trained immunity” with his colleagues back in 2011 to explain how BCG helps build stronger immunity against many more pathogens than Tuberculosis. Dr Netea also recently co-authored an article in The New England Journal of Medicine, “Trained Innate Immunity, Epigenetics, and COVID-19,” where he laid out the theory behind how BCG may be providing protection against COVID-19.

On December 11, the BCG PRIME team announced at the International Symposium for Innate Immunity and COVID-19 that they expected results by the end of January.
If BCG is proven to be effective against COVID-19, it is imperative to address the notion that BCG is what is protecting the elderly in countries with high rates of inoculation.
What really matters in the case of COVID-19 is whether the elderly in a country, i.e. those over 70 years of age, are protected or not. The countries where we found strong evidence that a majority or supermajority of the over-70 population have received BCG include Greece, Norway, Finland, Denmark, Germany, New Zealand, Australia, Japan, Vietnam and Uruguay. These countries have very low death rates per capita due to COVID-19, far lower than those observed in the US, Italy, Belgium, and the Netherlands, the only four nations in the world to have never adopted BCG.
In fact, Belgium has the highest COVID-19 death rate per capita of any country in the world, though it ranks 17th among all nations on the United Nation’s Human Development Index (HDI). Even more remarkable is that Greece, with a similar sized population to Belgium, has a death rate per capita that is one quarter that of Belgium, despite being ranked 32nd on the HDI, and with a far weaker healthcare system. Greece introduced BCG in 1925 and by the early 1950s had vaccinated a vast majority of the children under the age of 18, meaning a supermajority of the current 70+ population born in Greece is BCG vaccinated compared to almost no local born person over age 70 vaccinated in Belgium.
If the BCG PRIME trial shows proof of immunity against COVID-19, then critical decisions will need to be made to determine how to prioritize the new supply of BCG
Japan provides another useful example of a country where the correlation appears to indicate that there is likely causality. Having initially introduced BCG in 1924, Japan decided in 1949 to vaccinate all citizens under 30. Thus, we expect that almost everyone between 70-101 years of age in Japan has been vaccinated with BCG. This may explain why Japan, a country of 126 million people, with 27 million over the age of 70, has lost only 3,300 of its citizens due to COVID-19 in 2020, despite Japan’s initial “half-hearted” response to the pandemic.
All of the other countries that have high death rates per capita (UK, France, Spain, Brazil, Mexico, Chile) are those where we did not find any evidence of a majority of the population over 70 having BCG protection.
If the BCG PRIME trial shows proof of immunity against COVID-19, then critical decisions will need to be made to determine how to prioritize the new supply of BCG which we expect to be produced by the 20+ existing global manufacturers at a rapid pace.
When analyzing the effectiveness of BCG over a lifetime, it was observed in a clinical trial conducted between 1935 and 1998 among almost 3,000 indigenous American and Alaskan peoples that those who received BCG between 1935 and 1938 had a 60 percent lower chance of developing lung cancer 60 years later as adults, compared to those in the control placebo group, highlighting the possible long-term benefits of BCG.
While our research did not find evidence of high BCG coverage for the over-70 population in any Latin American country other than Uruguay, there are a large number of countries in Asia and Africa that have fared well during the COVID-19 crisis, even without heavy BCG coverage in the over 70 population, such as Pakistan, Malaysia, Nigeria, and Egypt.
While more research and trials need to be conducted to better understand why so many of these countries where the elderly are not covered with BCG tend to have very low mortality rates, our analysis leads us to attribute the positive outcomes in these countries to a high level of Latent Tuberculosis among the elderly.
Several recent studies, including an instrumental variable method analysis from Japan, highlight a strong correlation between low death rates from COVID-19 and high levels of Latent Tuberculosis in most Asian and African countries. According to these studies, Latin America has a much lower incidence of Latent Tuberculosis than Asia and Africa, closer to that of North America and Western Europe.

Our research also brought up an astonishing statistical disparity within the United States, where 38 percent of the roughly 378,000 deaths have taken place in nursing homes despite reflecting only five percent of cases. Homeless shelters, on the other hand, have been spared despite high levels of infections. In fact, in eight homeless shelters across the country with 2,665 residents, 778 people (29 percent) tested positive for COVID-19, yet not a single person died. In three of those homeless shelters, all 214 infected patients were asymptomatic, a near statistical impossibility given that an estimated 45 percent of COVID-19 patients are asymptomatic.
The US Center for Disease Controls (CDC) was reported to be actively looking into the “stunning” results from the Pine Street Inn Homeless shelter in Boston back in mid-April. While Latent Tuberculosis is quite rare in the US, prevalence of Latent TB in the homeless population in the United States has been reported to be as high as 67 percent, an incidence rate 20 times higher than the general population.
In his book, Discovering Tuberculosis, Christian McMillen writes that “the mass vaccination with BCG, is without doubt, the most far reaching and longest-lasting prophylactic medical campaign in world history.” According to McMillen, BCG, which was developed by Albert Calmette and Camille Guérin in 1921 at the Pasteur Institute in France, has been given to an estimated 4.5 billion people around the world, and another 1.8 billion carry Latent Tuberculosis, roughly 20x more people than those living with Active Tuberculosis disease. That means up to 6.3 billion people in the world could have the Tuberculosis germ either through the live attenuated Tuberculosis in the BCG vaccine or by virtue of infection with Latent Tuberculosis.
Our research and analysis suggest that countries with high levels of BCG vaccination and/or high levels of Latent Tuberculosis among the elderly have significantly lower mortality rates than those without. Many developed nations already have the new vaccines against COVID-19, some with contracts ample to vaccinate their populations several times over. If the results of the BCG PRIME trial are positive, the elderly in many developing countries, especially those in Latin America, who remain most at risk of COVID-19 fatality, should be prioritized in order to have a more equitable BCG rollout than has been the distribution of newly developed COVID-19 vaccines.
Kashif Zafar is a financial services executive based in New York and serves on the boards of the American Pakistan Foundation and Middlebury College.
Adil Alvi is a first year student at Middlebury College having graduated from The Lyceum in Karachi, Pakistan

